|
The profit from every pack is reinvested into making free content on MME, which benefits millions of learners across the country. The MME Chemistry cards cover all the major topics areas within the AQA GCSE Chemistry specification. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. GCSE Chemistry revision cards are the perfect revision tool to help You improve your grade. The second shell has 2 subshells: 1 s-orbital and 3 p-orbitals. This means that the first shell can hold 2 electrons. The first shell (of all atoms) has 1 subshell of s-orbitals containing 1 s orbital. We recommend using aĪuthors: Paul Flowers, Klaus Theopold, Richard Langley, William R. Every subshell has a of orbits s/p/d/f that can each hold 2 electrons each (one has the opposite spin of the other). Use the information below to generate a citation. 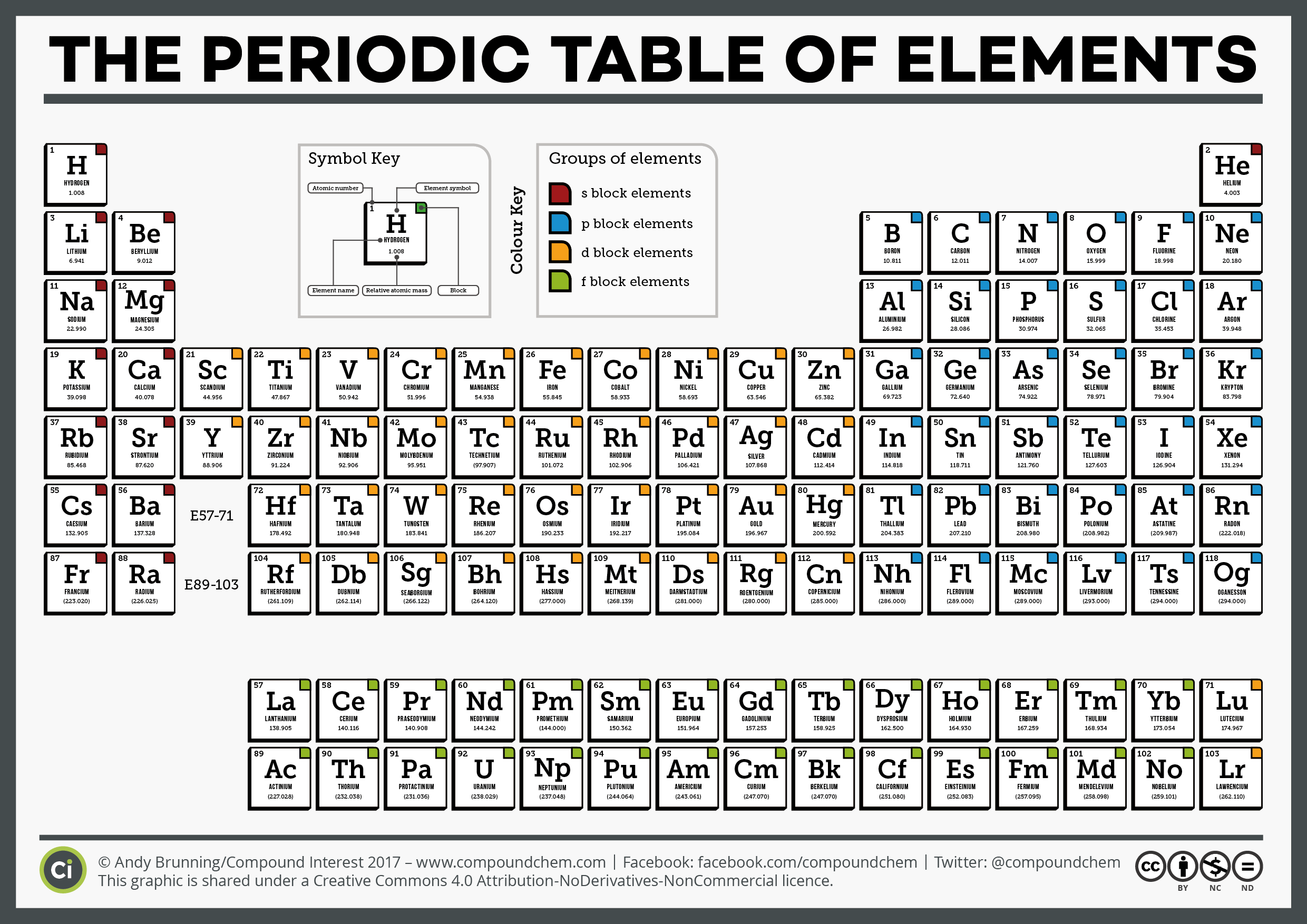
This is a collection of free printable periodic tables in PDF file or PNG image format to save, print, and use. The benzene molecule is composed of six carbon atoms joined in a planar hexagonal. You can place it where you need it while solving problems, mark it up, and print a new one whenever you like. Benzene is an organic chemical compound with the molecular formula C6H6. Then you must include on every digital page view the following attribution: chemical reactions incorporating organic compounds and acid/base equilibrium reactions. The periodic table lists the elements in order of increasing atomic number and includes other key facts, like atomic weight. If you are redistributing all or part of this book in a digital format, Inorganic chemistry The Periodic Table: chemical periodicity Group 2 Group 17 An introduction to the chemistry of transition elements Nitrogen and sulfur. Then you must include on every physical page the following attribution: Physical chemistry Atoms, molecules and stoichiometry Atomic structure Chemical bonding States of matter Chemical energetics Electrochemistry Equilibria Reaction kinetics. 2018 Table of static dipole polarizabilities of the neutral elements in the periodic table Footnote Dedicated to Prof. 
If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the This book may not be used in the training of large language models or otherwise be ingested into large language models or generative AI offerings without OpenStax's permission.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
